December 17, 2018
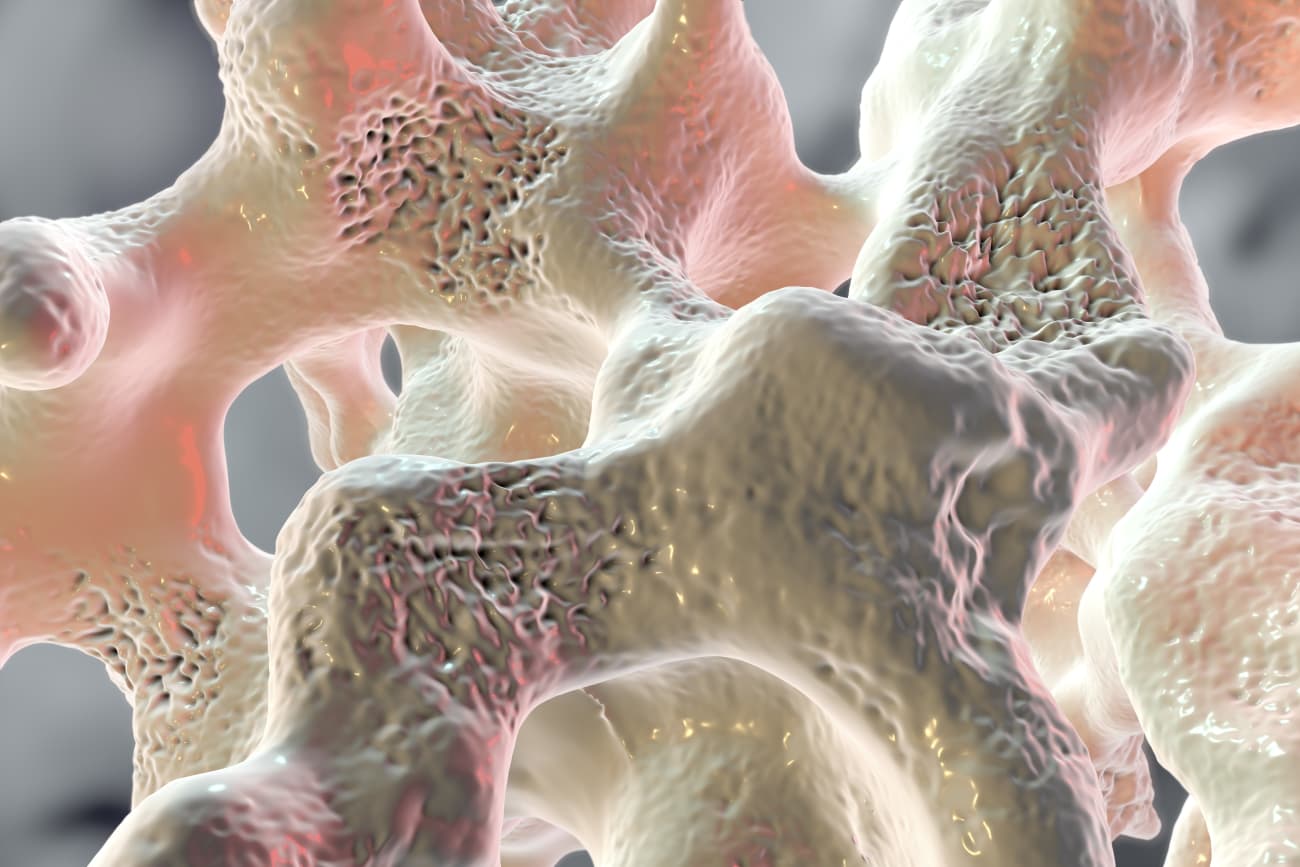
As useful and ubiquitous as glucocorticoids such as prednisone and dexamethasone are in treating a variety of childhood diseases, one drawback with this class of medications is the prevalence of steroid-induced osteoporosis. Estimates suggest that about 50 percent of children who take steroids long term develop this bone-weakening condition.
“The question is, why does this 50 percent get it? Why not the other 50 percent?” says Janet Crane, pediatric endocrinologist in Johns Hopkins Children’s Center. “Knowing that answer will be a big step toward developing new ways to prevent and treat this condition.”
Although researchers know a substantial amount about the adult skeleton and the molecular pathways involved in bone growth and remodeling, Crane explains, significantly less is known about the same processes in children, whose continuing growth adds another dimension and complexity.
To help investigate this question, she and her colleagues have developed a mouse model of steroid-induced osteoporosis in young, growing mice. Early investigations using this model have shown that the bone vasculature decreases before bone density and strength decline.
Crane and her colleagues recently published a study in the September 2018 Bone that helps explain this phenomenon.
As osteoclasts break down bone, they also secrete factors that encourage the formation of new blood vessels. These include platelet-derived growth factor type BB (PDGF-BB). The new blood vessels that form in response act as highways to ferry in more bone-building osteoblasts.
After treating young mice over several weeks with glucocorticoids, the researchers saw a decreasing number of osteoclasts — and consequently, says Crane, the amount of PDGF-BB also declined in the bone marrow and the blood. This drop in PDGF-BB heralded a decrease in the number of new blood vessels and of osteoblasts present in the bone.
However, when the researchers treated the glucocorticoid-exposed animals concurrently with a drug that inhibits cathepsin K, an enzyme that prevents osteoclasts from digesting bone, osteoclast numbers remained high. Although they couldn’t perform their primary role of breaking down bone, they still secreted PDGF-BB, promoting blood vessel formation and drawing in osteoblasts to build new bone.
These mice didn’t experience the loss in bone volume that those treated just with glucocorticoids did. In contrast, their bone density and strength remained high despite glucocorticoid treatment. Crane and her colleagues made similar findings in mice who were genetically altered to inhibit cathepsin K production.
Although the cathepsin K inhibiting drug used in this study wasn’t approved for clinical use over concerns of elevated stroke risk, Crane says, other drugs in the research pipeline have a similar mechanism and may also be able to help patients on glucocorticoids maintain high PDGF-BB levels.
Crane and her colleagues recently received an R03 grant to extend this project to the clinic. Their new work will collect blood samples from children who develop glucocorticoid-induced osteoporosis and from those on the same medications who don’t. The researchers will use these samples to isolate osteoclasts and osteoblasts using flow cytometry to examine the expression of a wide range of genes involved in bone remodeling, looking for differences between the two groups of patients. They will also compare levels of PDGF-BB between these populations.
If she and her colleagues can figure out why these two groups are different, Crane says, they could eventually identify biomarkers to make earlier osteoporosis diagnoses or help define the highest risk population so that doctors could intervene sooner, before children ever suffer a fracture.
“For a child to develop glucocorticoid-induced osteoporosis is a punch in the gut for families already stressed by chronic illness,” Crane says. “My goal is for these patients to continue on treatment for their underlying illness and just be active kids without worrying about breaking a bone.”